The Kinesia 360™ motor assessment system provides low burden method for remote, continuous measurement of patient symptoms. Sensors worn on the wrist and ankle combined with a mobile application continuously record data for assessment of tremor, slowness, dyskinesia and mobility. Data from the motion sensors is uploaded to the Kinesia Web Portal and algorithms are used to detect symptoms and calculate severity scores every two minutes on a scale shown to be highly correlated with clinician ratings.
Kinesia 360
Continuous Ambulatory Monitoring for Parkinson’s Disease and Other Movement Disorders
Continuous ambulatory monitoring
Real-time data analysis & reporting
Measure with high sensitivity & reliability
The Kinesia 360 motor assessment system provides low burden method for remote, continuous measurement of patient symptoms. Sensors worn on the wrist and ankle combined with a mobile application continuously record data for assessment of tremor, slowness, dyskinesia and mobility. Data from the motion sensors is uploaded to the Kinesia Web Portal and algorithms are used to detect symptoms and calculate severity scores every two minutes on a scale shown to be highly correlated with clinician ratings.
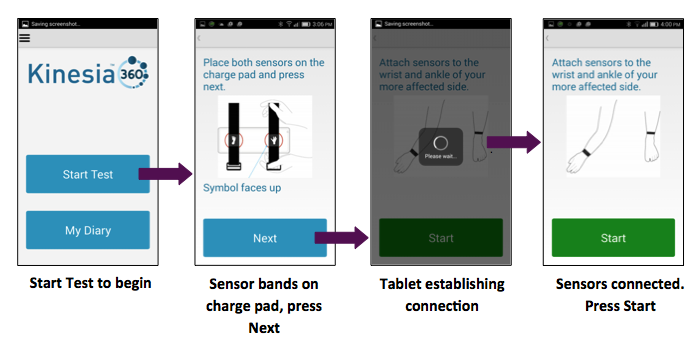
The Sensors
The Kinesia 360 user kit includes two sensors, one worn on the wrist and one on the ankle, to allow an accurate and clinically validated assessment of Parkinson’s motor symptoms. Sensors record data all day and recharge overnight for extended home use.
Kinesia 360 Smartphone App
The smartphone is preloaded with the Kinesia 360 software app and guides patients through daily use. The app also includes electronic diaries for capturing patient-reported outcomes and customizable medication diaries. The system also includes mobile device management to ensure software is always current. Kinesia 360 software has been translated into over 20 languages and dialects and may be translated into additional languages upon request.
Secure Web Portal
Motion and diary data are transmitted to a secure cloud via mobile broadband for processing and storage in a 21 CFR Part 11 compliant database. Clinicians and researchers can access reports via the Kinesia web portal and view real-time patient compliance.
Kinesia 360 Reports
Kinesia 360 provides motor symptom data every two-minutes as well as daily summaries. Customized data reporting is available.
Continuous Outcome Measures
- Tremor
- Slowness
- Dyskinesia
- Mobility
- Steps
- Body position
Click HERE for a complete list of clinical validation publications.
Kinesia™ products are used as outcome measures in Phase I, II, III and IV clinical trials.
Kinesia technology is integrated in clinical trials around the globe for Parkinson’s disease, essential tremor and other movement disorders. Intelligent remote sensing technology increases the sensitivity and reliability of outcome measures, improves efficiency with web applications and expands accessibility. To view publications, please click HERE.
